Coordination CompoundHard
Question
A solution containing 2.675 g of CoCl3.6 NH3 (molar mass = 267.5 g mol-1) is passed through a cation exchanger. The chloride ions obtained in solution were treated with excess of AgNO3 to give 4.78 g of AgCl (molar mass = 143.5 g mol-1). The formula of the complex is (At. Mass of Ag = 108 u)
Options
A.[Co(NH3)6]Cl3
B.[CoCl2(NH3)4]Cl
C.[CoCl3(NH3)3]
D.[CoCl(NH3)5]Cl2
Solution
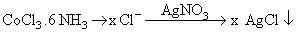
n(AgCl) = x n(CoCl3. 6NH3)
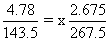 ∴ x = 3
∴ x = 3∴ The complex is [Co(NH3)6]Cl3
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The coordination number of central metal atom in a complex is determined by...Which of the following statements is correct?...Aqueous solution of FeSO4 gives tests for both Fe2+ and SO42– but after addition of excess of KCN, solution ceases to gi...The geometry of [NiCI4]2- and [NiCI2 (PMe3)2] are :...The EAN of metal atoms in Fe(CO)2(NO)2 and Co2(CO)8 respectively are...