Coordination CompoundHard
Question
Which one of the following is an outer orbital complex and exhibits paramagnetic behaviour ?
Options
A.[ Ni(NH3)6 ]2+
B.[ Zn(NH3)6 ]2+
C.[ Cr(NH3)6 ]3+
D.[ Co(NH3)6 ]3+
Solution
Outer orbital complex utilises nd orbitals for bonding and exhibit paramagnetic behaviour, only if there present unpaired electrons.
(a) In [ Ni ( NH3)6]2+:
Ni2+ = [Ar] 3d8 4s0
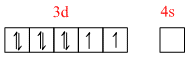
[Ni(NH3)6]2+ =
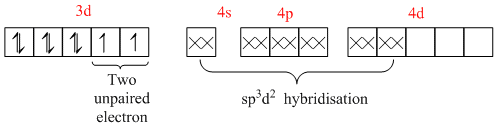
So, this is an outer orbital complex having paramagnetic character.
(b) In [ Zn ( NH3)6]2+ :
Zn2+ = [ Ar ] 3d10
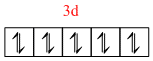
[Zn(NH3)6]2+ =
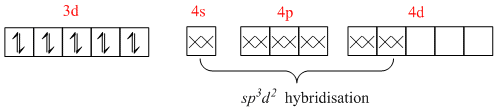
Thus, it is also an outer orbital complex but it is dimagnetic as all the electrons are paired.
(C) In [ Cr(NH3)6]3+ :
Cr3+ = [Ar]3d3
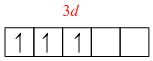
[Cr( NH3)63+ =
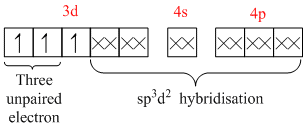
Because of the involvement of (n1)d, i.e., 3d orbital in hybridisation, it is an inner orbital complex. Its nature is paramagnetic because of the presence of three unpaired electrons.
(d) In [ Co( NH3)6]3+ :
Co3+ = [ Ar ] 3d6
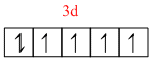
[Co(NH3)6]3+ =
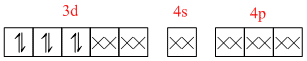
As all the electrons are paired it is dimagnetic complex.
(a) In [ Ni ( NH3)6]2+:
Ni2+ = [Ar] 3d8 4s0

[Ni(NH3)6]2+ =

So, this is an outer orbital complex having paramagnetic character.
(b) In [ Zn ( NH3)6]2+ :
Zn2+ = [ Ar ] 3d10

[Zn(NH3)6]2+ =

Thus, it is also an outer orbital complex but it is dimagnetic as all the electrons are paired.
(C) In [ Cr(NH3)6]3+ :
Cr3+ = [Ar]3d3

[Cr( NH3)63+ =

Because of the involvement of (n1)d, i.e., 3d orbital in hybridisation, it is an inner orbital complex. Its nature is paramagnetic because of the presence of three unpaired electrons.
(d) In [ Co( NH3)6]3+ :
Co3+ = [ Ar ] 3d6

[Co(NH3)6]3+ =

As all the electrons are paired it is dimagnetic complex.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following has an optical isomer ?...The value of the ′spin only′ magnetic moment for one of the following configurations is 2.84 BM. The correct...A white precipitate of AgCl dissolves in NH4OH due to the formation of...Among the following pairs of ions, the lower oxidation state in aqueous solution is more stable than the other, in...π-bonding is not involved in:...