Coordination CompoundHard
Question
Which of the following statements is/are true for the complexes, [Fe(H2O)6]2+, [Fe(CN)6]4-, [Fe(C2O4)3]3- and [Fe(CO)5] ?
Options
A.Only [Fe(C2O4)3]3- show optical isomerism.
B.[Fe(C2O4)3]3- is less stable than [Fe(CN)6]2-
C.All complexes have same effective atomic number.
D.[Fe(CO)5] shows back bonding.
Solution
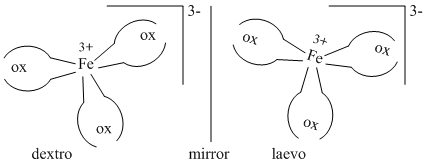
[Fe(H2O)6]2+ and [Fe(CN)6]4- do not show optical isomerism because of the presence of plane of symmetry as well as centre of symmetry.
(B) [Fe(C2O4)3]3 has three, five membered chelating rings and thus is more stable due to chelation.
(C) [Fe(H2O)6]2+ ; EAN = 26 - 2 + 12 = 36
[Fe(CN)6]4- ; EAN = 26 - 2 + 12 = 36
[Fe(C2O4)3]3- ; EAN = 26 - 3 + 12 = 35
(D) Fe
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
In the complex [CoCl2(en)2]Br, the co-ordination number and oxidation state of cobalt are :...Which of the following is not correctly matched ?...A solution containing 2.675 g of CoCl3.6 NH3 (molar mass = 267.5 g mol-1) is passed through a cation exchanger. The chlo...An ion M2+, forms the complexes [M(H2O)6]2+, [M(en)3]2+ and [MBr6]4–, match the complex with the appropriate colour....What is incorrect about homoleptic metal carbonyls?...