Coordination CompoundHard
Question
Which of the following isomers of [M(NH3)2Cl2] would react with silver oxalate (Ag2C2O4) ?
Options
A.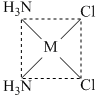

B.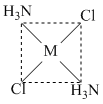

C.Both
D.None
Solution
It is polar in nature. The chelate rings require the cis structure, because the chelating ligand is too small to span the trans positions.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Cis-trans isomerism is found in square planar complexes of molecular formula (′a′ and ′b′ are mo...The correct IUPAC name of the complex:...Consider the following statements ;(I) [Mn(H2O)4]SO4 is paramagnetic and square planar.(II) Crystal field splitting ener...EAN of a metal carbonyl M(CO)x is 36. If atomic number of metal M is 26, what is the value of x ?...What is the charge on the complex [Cr(C2O4)2(H2O)2] formed by Cr(III) ?...