Coordination CompoundHard
Question
Which of the following is/are inner orbital complex(es) as well as dimagentic in nature ?
Options
A.[Zn(NH3)6]2+
B.[Ni(NH3)6]2+
C.[Cr(NH3)6]3+
D.[Co(NH3)6]3+
Solution
(A) Because of 3d10 configuration no (n - 1)d orbital is available for d2sp3 hybridisation and thus forms outer orbital complex. The complex is diamagnetic.
(B) Because of 3d8 configuration no (n - 1)d orbital is available for d2sp3 hybridisation and thus forms outer orbital complex. The complex is paramagnetic with two unpaired electrons.
(C) The complex is inner orbital complex but 3d3 configuration has three unpaired electrons with weak as well as with strong field ligand.
(D) In the diamagnetic octahedral complex, [Co(NH3)6]3+, the cobalt ion is in +3 oxidation state and has the electronic configuration represented as shown below.
Co3+,[Ar]3d6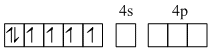
[Co(NH3)6]3+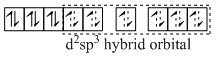
(inner orbital or d2sp3 hybrid orbital
low spin complex) Six pairs of electrons from six NH3 molecules.
(B) Because of 3d8 configuration no (n - 1)d orbital is available for d2sp3 hybridisation and thus forms outer orbital complex. The complex is paramagnetic with two unpaired electrons.
(C) The complex is inner orbital complex but 3d3 configuration has three unpaired electrons with weak as well as with strong field ligand.
(D) In the diamagnetic octahedral complex, [Co(NH3)6]3+, the cobalt ion is in +3 oxidation state and has the electronic configuration represented as shown below.
Co3+,[Ar]3d6
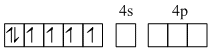
[Co(NH3)6]3+
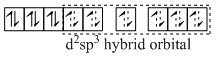
(inner orbital or d2sp3 hybrid orbital
low spin complex) Six pairs of electrons from six NH3 molecules.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The number of unpaired electrons in d6, low spin, octahedral complex is :...Which one of the following statements concerning lanthanide elements is false ?...Given below are two statements:Statement I: Crystal Field Stabilization Energy (CFSE) of $\left\lbrack Cr\left( H_{2}O \...Which of the following statements is incorrect ?...What is incorrect about homoleptic metal carbonyls?...