Coordination CompoundHard
Question
If excess of AgNO3 solution is added to 100 mL of a 0.024 M solution of dichlorobis(ethylenediamine)cobalt (III) chloride. How many moles of AgCl be precipitated ?
Options
A.0.0012
B.0.0016
C.0.0024
D.0.0048
Solution
[CoCl2(en)2]Cl, One mole complex contains, one mole of ionisable Cl-.
One mole of complex = one mole of Cl-.
∴ One mole of AgCl = One mole of complex
=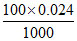 = 0.0024.
= 0.0024.
One mole of complex = one mole of Cl-.
∴ One mole of AgCl = One mole of complex
=
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The complex [Pt(NH3)4]2+ has ..... structure :...Given that the energy of the photons of different colours decreases in the order of v i b g y o r (Violet > Indigo &g...[Fe(en)2(H2O)2]2+ + en → complex(X). The correct statement about the complex (X) is :...The value of the ′spin only′ magnetic moment for one of the following configurations is 2.84 BM. The correct...Low spin complex is formed by :...