Coordination CompoundHard
Question
Which of the following complexes is correctly matched with their geometries ?
Options
A.[Co(py)4]2+ - square planar
B.[Cu(CN)4]3- - tetrahedral
C.[Fe(CO)4]2- - tetrahedral
D.All of these
Solution
(A) Py is strong field ligand and the complex is square planar and paramagnetic.
(B) Copper (I) has electron configuration [Ar]18 3d10. With CN- (strong field ligand) there is no empty d-orbital for dsp2 hybridisation and so the complex is tetrahedral not square planar.
(C) Iron is in -2 oxidation state and CO is strong field ligand so after rearrangement of electrons, pairing of electrons occurs. So,
[Fe(CO)4]2-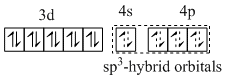
(B) Copper (I) has electron configuration [Ar]18 3d10. With CN- (strong field ligand) there is no empty d-orbital for dsp2 hybridisation and so the complex is tetrahedral not square planar.
(C) Iron is in -2 oxidation state and CO is strong field ligand so after rearrangement of electrons, pairing of electrons occurs. So,
[Fe(CO)4]2-

Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Consider the following statementsS1 : [ Cr (NH3)6]3+ is a inner orbital complex with crystal field stabilization energy ...Formula of ferrocene is:...Which of the following is not onsidered as an organometallic compound ?...Both geometrical and optical isomerism are shown by :...Consider the following statements: According the Werner's theory....