Coordination CompoundHard
Question
Consider the following statements
S1 : [ Cr (NH3)6]3+ is a inner orbital complex with crystal field stabilization energy equal to - 1.2 Do
S2 : The complex formed by joining the CN- ligands to Fe3+ ion has theoretical value of ′spin only′ magnetic moment equal to 1.73 B.M.
S3 : Na2S + Na2 [Fe (CN)5 NO] → Na4 [ Fe (CN)5 NOS], In reactant and product the oxidation states of iron are sameand arrange in the order of true/false.
S1 : [ Cr (NH3)6]3+ is a inner orbital complex with crystal field stabilization energy equal to - 1.2 Do
S2 : The complex formed by joining the CN- ligands to Fe3+ ion has theoretical value of ′spin only′ magnetic moment equal to 1.73 B.M.
S3 : Na2S + Na2 [Fe (CN)5 NO] → Na4 [ Fe (CN)5 NOS], In reactant and product the oxidation states of iron are sameand arrange in the order of true/false.
Options
A.F T F
B.T T F
C.T T T
D.F F F
Solution
S1 : Cr3+  CFSE = 3 × - 0.4 = - 1.2 ᐃ0, hybridisation is d2sp3 (NH3 is strong ligand)
CFSE = 3 × - 0.4 = - 1.2 ᐃ0, hybridisation is d2sp3 (NH3 is strong ligand)
S2 : Fe3+, 3d5 - one unpaired electron after pairing (CN- is stronger field ligand)
∴ μ =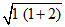 ≈ 1.73 BM
≈ 1.73 BM
S3 : [FeII(CN-5)5NO+]2- and [FeII(CN-5)5NOS-1]1-
In reactant and product, the iron is in same oxidation state i.e. +2.
 CFSE = 3 × - 0.4 = - 1.2 ᐃ0, hybridisation is d2sp3 (NH3 is strong ligand)
CFSE = 3 × - 0.4 = - 1.2 ᐃ0, hybridisation is d2sp3 (NH3 is strong ligand)S2 : Fe3+, 3d5 - one unpaired electron after pairing (CN- is stronger field ligand)
∴ μ =
S3 : [FeII(CN-5)5NO+]2- and [FeII(CN-5)5NOS-1]1-
In reactant and product, the iron is in same oxidation state i.e. +2.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which method can be used to distinguish [Co(NH3)6][Cr(NO2)6] and [Cr(NH3)6][Co(NO2)6]...The most stable ion is...Which of the following has five donor (coordinating) sites?...$\left\lbrack Ni\left( {PPh}_{3} \right)_{2}{Cl}_{2} \right\rbrack$ is a paramagnetic complex. Identify the INCORRECT st...For the reaction, Ni2+ + 4NH3 ⇋ [Ni(NH3)4]2+ at equilibrium, if the solution contains 1.6 ×10-4% of nickel in...