Coordination CompoundHard
Question
K6[(CN)5 CO - O - O - CO(CN)5] 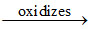 K5[(CN)5Co - O - O - Co(CN)5]
K5[(CN)5Co - O - O - Co(CN)5]
(X) (Y)
In both the complexes Co have t2g6eg0 configuration.
The bond energy of (O - O) in X and Y is :
(X) (Y)
In both the complexes Co have t2g6eg0 configuration.
The bond energy of (O - O) in X and Y is :
Options
A.bond energy of (O - O) in Y < bond energy of (O - O) in X.
B.bond energy of (O - O) in X < bond energy of (O - O) in Y.
C.bond energy of (O - O) in X = bond energy of (O - O) in Y.
D.bond energy of (O - O) in X and bond energy of (O - O) in Y can′t be comparable.
Solution
K6[(CN-5)5 Co (III) - O - O - Co (III) (CN-5)5]6+ 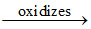 K5[(CN-5)5 Co - O-1 - Co(CN-5)5]5+.
K5[(CN-5)5 Co - O-1 - Co(CN-5)5]5+.
O22-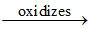 O2- ; bond order of O22- =
O2- ; bond order of O22- = 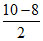 = 1; bond order of O2- =
= 1; bond order of O2- = 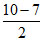 = 1.5
= 1.5
Bond strength ∞ bond order ; so,
Bond energy of O22- < Bond energy of O2-.
O22-
Bond strength ∞ bond order ; so,
Bond energy of O22- < Bond energy of O2-.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
When AgNO3 is added to a solution of Co(NH3)5Cl3, the precipitante of AgCI shows two ionizable chloride ions. This mean:...Which of the following is a high spin complex ?...Ethylene diamine is an example of a .......... ligand :...Which of the following statements is(are) correct ?...Which of the following is considered to be an anticancer species?...