Coordination CompoundHard
Question
Which of the following statements is(are) correct ?
Options
A.[Ag(NH3)2]+ is linear with sp hybridised Ag+ ion.
B.NiCl42-, VO43- and MnO4- have tetrahedral geometry.
C.[Cu(NH3)4]2+, [Pt(NH3)4]2+ and [Ni(CN)4]2- have dsp2 hybridisation of the metal ions.
D.Fe(CO)5 have trigonal bipyramidal structure with dsp3 hybridised iron.
Solution
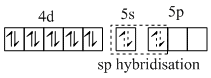
(A) [Ag(NH3)2]-
Ag(I) → 4d10 5s0 H2N → Ag → NH3
Liner
(B) NiCl42-, VO43- and MnO4- have tetrahedral geometries.
Ni2+ → 3d8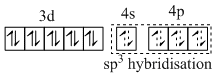
(C) [Cu(NH3)4]2+, [Pt(NH3)4]2+ and [Ni(CN)4]2- have square planar geometries.
[Cu(NH3)4]2+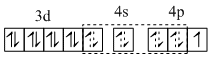
Pt2+, [Xe]4f14 5d8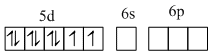
[Pt(NH3)4]2+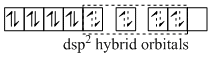
Similarly the hybridisation scheme for [Ni(CN)4]2- (CN- is a strong field ligand) is as shown in figure.
Ni2+, [Ar]3d8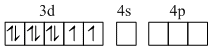
[Pt(NH3)4]2+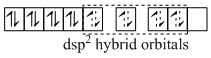
(D) [Fe(CO)5] is diamagnetic and has dsp3 hybridisation. So it has trigonal bipyramidal geometry.
Ag(I) → 4d10 5s0 H2N → Ag → NH3
Liner

(B) NiCl42-, VO43- and MnO4- have tetrahedral geometries.
Ni2+ → 3d8
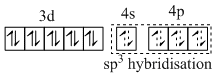
(C) [Cu(NH3)4]2+, [Pt(NH3)4]2+ and [Ni(CN)4]2- have square planar geometries.
[Cu(NH3)4]2+

Pt2+, [Xe]4f14 5d8

[Pt(NH3)4]2+

Similarly the hybridisation scheme for [Ni(CN)4]2- (CN- is a strong field ligand) is as shown in figure.
Ni2+, [Ar]3d8
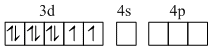
[Pt(NH3)4]2+

(D) [Fe(CO)5] is diamagnetic and has dsp3 hybridisation. So it has trigonal bipyramidal geometry.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following is correct about? Tetraamminedithiocyanato-scobalt(III) tris(oxalato)cobaltate(III)...Which amongst the following are organometallic compounds ?1. Al2(CH3)62. K[PtCl3C2H2]3. N(CH3)3...The pair(s) of coordination complex/ion exhibiting the same kind of isomerism is(are) -...Which one of the following complexes exhibit chirality ?...The correct IUPAC name for the compound [Co(NH3)4CI(ONO)]Cl is :...