MetallurgyHard
Question
Select the correct statement :
Options
A.Dolomite contains both magnesium and calcium.
B.Extraction of lead from galena involves roasting in limited supply of air at moderate temperature followed by self reduction at higher temperature (to melt the charge).
C.Extraction of zinc from zinc blende involves roasting followed by reduction with carbon.
D.The chemical composition of ′slag′ formed during the extraction of iron and copper is FeSiO3.
Solution
(A) dolomite is CaCO3.MgCO3
(B) 2PbS + 3O2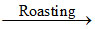 2PbO + 2SO2
2PbO + 2SO2
PbS + 2PbO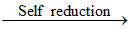 3Pb + SO2
3Pb + SO2
(C) 2ZnS + 3O2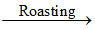 2ZnO + 2SO2
2ZnO + 2SO2
ZnO + C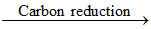 Zn + CO
Zn + CO
(D) In extraction of iron the slag obtained is CaSiO3 where as in copper it is FeSiO3.
(B) 2PbS + 3O2
PbS + 2PbO
(C) 2ZnS + 3O2
ZnO + C
(D) In extraction of iron the slag obtained is CaSiO3 where as in copper it is FeSiO3.
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
Silver ore dissolves in dilute solution of NaCN in the presence of air to form :...In which of the following extration no reducing agent is required ?...In which of the following isolations no reducing agent is required:...Consider the following statements : S1 : Poling process is used for the refining of copper and lead. S2 : The scavenger ...Which one of the following reactions is an example of calcination process ?...