MetallurgyHard
Question
Which one of the following reactions is an example of calcination process ?
Options
A.2 Ag + 2HCl + [O] → 2 AgCl + H2O
B.2 Zn + O2 → 2 ZnO
C.2 ZnS + 3O2 → 2 ZnO + 2 SO2
D.MgCO3 → MgO + CO2
Solution
Conversion of a carbonate into oxide is an example of calcination
MgCO3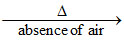 MgO + CO2 ↑.
MgO + CO2 ↑.
MgCO3
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
Froth floatation process for the concentration of sulphide ores is an illustration of the practical application of:...Extraction of zinc from zinc blende is achieved by...Which of the following reaction (s) occurs during calcination?...Which of the following reaction(s) occur during calcination?...Which of the following is / are correctly matched ?...