MetallurgyHard
Question
In which of the following extration no reducing agent is required ?
Options
A.Iron from haematite.
B.Aluminium from bauxite.
C.Magnesium from carnallite.
D.Zinc from zinc blende
Solution
(A) 3Fe2O3 + CO → 2Fe3O4 + CO2 ; Fe3O4 + CO → 3FeO + CO2 ; FeO + CO → Fe + CO2
(B) The purified Al2O3 is mixed with Na3AlF6 (cryolite) or CaF2 (fluorspar) ) which lowers the melting point of the mixture and increases conductivity. The fused matrix is electrolysed.
Cathode : Al3+ (melt) + 3e- Al(l)
Anode : C(s) + O2- (melt) CO(g) + 2e-
C(s) + 2O2- (melt) CO2 (g) + 4e-
Electrons act as reducing agent in electrolytic reduction.
(C) The molten mixture containing MgCl2, NaCl and CaCl2 is electrolysed.
MgCl2 ⇋ Mg2+ + 2Cl-
At cathode : Mg2+ + 2e- Mg(99% pure) ;
At anode : 2Cl- Cl2 + 2e-
Electrons act as reducing agent in electrolytic reduction.
(D) 2ZnS + 3O2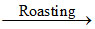 2ZnO + 2SO2
2ZnO + 2SO2
ZnO + C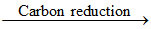 Zn + CO
Zn + CO
(B) The purified Al2O3 is mixed with Na3AlF6 (cryolite) or CaF2 (fluorspar) ) which lowers the melting point of the mixture and increases conductivity. The fused matrix is electrolysed.
Cathode : Al3+ (melt) + 3e- Al(l)
Anode : C(s) + O2- (melt) CO(g) + 2e-
C(s) + 2O2- (melt) CO2 (g) + 4e-
Electrons act as reducing agent in electrolytic reduction.
(C) The molten mixture containing MgCl2, NaCl and CaCl2 is electrolysed.
MgCl2 ⇋ Mg2+ + 2Cl-
At cathode : Mg2+ + 2e- Mg(99% pure) ;
At anode : 2Cl- Cl2 + 2e-
Electrons act as reducing agent in electrolytic reduction.
(D) 2ZnS + 3O2
ZnO + C
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
In the metallurgy of iron, the upper layer obtained in the bottom of blast furnace mainly contains :...Which of the following is(are) sulphide ores?...Magnetic separation process may be used for the concentration of :...Hydrogen has high ionization energy than alkali metals because it has :...Process of heating ore in air to remove sulphur is :...