Atomic StructureHard
Question
Starting with a simple of pure 66Cu, 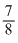 of it decays in to Zn in 15 minutes. The corresponding half life is
of it decays in to Zn in 15 minutes. The corresponding half life is
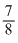 of it decays in to Zn in 15 minutes. The corresponding half life is
of it decays in to Zn in 15 minutes. The corresponding half life isOptions
A.15 minutes
B.10 minutes
C.7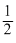 minutes
minutes
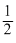 minutes
minutesD.5 minutes
Solution
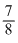 days of Cu decays.
days of Cu decays.∴ Cu undecayed, N = 1 -
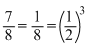
∴ No. Of half life = 3
⇒ half life period, T =
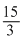 = 5 minutes
= 5 minutesCreate a free account to view solution
View Solution FreeMore Atomic Structure Questions
Radioactive nuclei that are injected into a potient collected at certain sites within its body, undergoing radioactive d...As per Bohr's model, the minimum energy (in eV) required to remove an electron from the ground state of doubly ionized L...Activity of a radioactive sample decreases to (1/3)rd of its original value in 9 days its activity will become...The volume (V) of a monatomic gas varies with its temperature (T), as shown in the graph. The ratio of work done by the ...The dependence of binding energy per nucleon, Î’N, on the mass number A, is represented by...