Analytical ChemistryHard
Question
Ammonium salts on heating with slaked lime liberates a colourless gas (X). Identify the correct statement for gas (X).
Options
A.(X) turns red litmus blue and produces dense white fumes in contact with dilute HCl.
B.(X) turns filter paper moistened with mercurous nitrate black and gives intense blue coloured solution with CuSO4(aq).
C.(X) when passed through Nessler′s reagent produces a brown colour precipitate.
D.All of these.
Solution
NH4+ + OH- 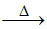 NH3 + H2O.
NH3 + H2O.
(A) NH3, alkaline in nature turns red litmus blue;
NH3 + HCl NH4Cl (white fumes)
(B) 2Hg NO3 + 2NH3 →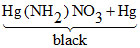 + NH4NO3 ; CuSO4 + 4NH3 → [Cu(NH3)4] SO4 (intense blue)
+ NH4NO3 ; CuSO4 + 4NH3 → [Cu(NH3)4] SO4 (intense blue)
(C) 2K2 (HgI4) + NH3 + 3KOH → HgOHgNH2I ↓ (brown) + 7KI + 2H2O
(A) NH3, alkaline in nature turns red litmus blue;
NH3 + HCl NH4Cl (white fumes)
(B) 2Hg NO3 + 2NH3 →
(C) 2K2 (HgI4) + NH3 + 3KOH → HgOHgNH2I ↓ (brown) + 7KI + 2H2O
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
Ni + H2SO4 (hot and concentrated) → X(g). The liberated gas (select the correct statement) :...When chlorine water is added to an aqueous solution of potassium halide in the presence of chloroform, a colour is devel...Select the incorrect statement....Consider the following statements, S1 : Hg2Cl2 precipitate undergoes disproportionation with ammonia solution. S2 : Hg2l...Aqueous Solution of BaBr2 gives yellow precipitate with :...