Analytical ChemistryHard
Question
Consider the following statements,
S1 : Hg2Cl2 precipitate undergoes disproportionation with ammonia solution.
S2 : Hg2l2 precipitate with excess I- ions undergoes disproportionation.
S3 : bismuth salt solution (chlorides or nitrates) produces a white precipitate when water is added to it in larger volume.
S4 : white precipitate of Cu(I) iodide produced by the reaction of CuSO4 and KI is intensely brown because of the formation of tri-iodide ions.
and arrange in the order of true / false.
S1 : Hg2Cl2 precipitate undergoes disproportionation with ammonia solution.
S2 : Hg2l2 precipitate with excess I- ions undergoes disproportionation.
S3 : bismuth salt solution (chlorides or nitrates) produces a white precipitate when water is added to it in larger volume.
S4 : white precipitate of Cu(I) iodide produced by the reaction of CuSO4 and KI is intensely brown because of the formation of tri-iodide ions.
and arrange in the order of true / false.
Options
A.T F T T
B.T T F F
C.T T T T
D.T F T F
Solution
S1 : Hg22+ + 2Cl- + 4NH3 + H2O → HgO.Hg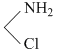 + Hg↓ + 3NH4Cl
+ Hg↓ + 3NH4Cl
S2 : Hg2I2 + 2I- → [HgI4]2- + Hg↓
S3 : BiCl3 + H2O → BiO+Cl- ↓ + 2HCl
S4 : Cu2+ + 5I- → 2CuI-↓ (white)
 + Hg↓ + 3NH4Cl
+ Hg↓ + 3NH4ClS2 : Hg2I2 + 2I- → [HgI4]2- + Hg↓
S3 : BiCl3 + H2O → BiO+Cl- ↓ + 2HCl
S4 : Cu2+ + 5I- → 2CuI-↓ (white)
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
CrO42- + H+ + H2O2 X + H2OIdentify the correct statement with respect to X....Which of the following statements is/are incorrect ?...CoS (black) obtained in group IV of salt analysis is dissolved in aqua regia and is treated with an excess of NaHCO3 and...When NH4Cl is added to a solution of NH4OH :...Ni + H2SO4 (hot and concentrated) → X(g). The liberated gas (select the correct statement) :...