Analytical ChemistryHard
Question
Select the incorrect statement.
Options
A. Marsh′s test involves the reduction of soluble arsenic compound to arsine by nascent hydrogen in acidic solution and its subsequent decomposition into hydrogen and metallic arsenic as a brownish-black mirror on heating in a glass tube mixed with hydrogen.
B.Ammonical silver nitrate gives metallic silver mirror with saturated solution of hydrazine sulphate.
C.Red precipitate of silver (I) chromate is soluble in dilute nitric acid and ammonia solution.
D.None of these
Solution
(A) As3+ + 3Zn + 3H+ → AsH3↑ + 3Zn2+ ; 4AsH3 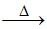 4As ↓ + 6H2 ↑
4As ↓ + 6H2 ↑
(B) 4[Ag(NH3)2]+ + H2N - NH2.H2SO4 → 4Ag↓ + N2 ↑ + 6NH4+ + 2NH3 ↑ + SO42-
(C) 2Ag2CrO4 ↓ + 2H+ ⇋ 4Ag+ + Cr2O72- + H2O
Ag2CrO4 ↓ + 4NH3 ⇋ 2[Ag(NH3)2]+ + CrO42-
So, all statements are correct.
(B) 4[Ag(NH3)2]+ + H2N - NH2.H2SO4 → 4Ag↓ + N2 ↑ + 6NH4+ + 2NH3 ↑ + SO42-
(C) 2Ag2CrO4 ↓ + 2H+ ⇋ 4Ag+ + Cr2O72- + H2O
Ag2CrO4 ↓ + 4NH3 ⇋ 2[Ag(NH3)2]+ + CrO42-
So, all statements are correct.
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
Consider the following statements, S1 : sulphide does give yellow precipitate with Cd(NO3)2 in aqueous solution. S2 : aq...Which of the following reagents gives a yellow precipitate with a hot faintly acidic solution of Bi3+ ions ?...Ni + H2SO4 (hot and concentrated) → X(g). The liberated gas (select the correct statement) :...An aqueous solution of salt gives white precipitate with AgNO3 solution as well as with dilute H2SO4. It may be...When CS2 layer containing both Br2 and I2 (2 : 1) is shaken with excess of chlorine (Cl2) water, the violet colour due t...