Analytical ChemistryHard
Question
Which of the following statement(s) is/are correct ?
Options
A.Yellow precipitated of silver arsenite is soluble in both nitric acid and ammonia.
B.Potassium cyanide when added in very small quantity to copper sulphate solution, produces first yellow precipitate which quickly converts in to white precipitate.
C.Black precipitate of BiI3 turns orange on heating with water.
D.White precipitate of Bi(OH)3 turns yellowish brown, when boiled.
Solution
(A) Correct.
(B) Cu2+ + 2CN- → Cu(CN)2↓ (yellow)
2Cu(CN)2↓ → 2Cu(CN)2↓ (white) + (CN)2 ↑
(C) BiI3↓ + H2O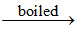 BiOI ↓ (orange) + 2HI
BiOI ↓ (orange) + 2HI
(D) Bi(OH)3↓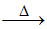 BiO.OH↓ (yellowish white) + H2O
BiO.OH↓ (yellowish white) + H2O
(B) Cu2+ + 2CN- → Cu(CN)2↓ (yellow)
2Cu(CN)2↓ → 2Cu(CN)2↓ (white) + (CN)2 ↑
(C) BiI3↓ + H2O
(D) Bi(OH)3↓
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
A white sodium salt dissolves in water to give a solution which is neutral to litmus. When silver nitrate solution is ad...Chromyl chloride vapours are dissolved in water and acetic acid and barium acetate solution is added, then:...Which of the following compound is formed in borax bead test ?...A mixture of two colourless substances was dissolved in water. When gaseous Cl2 was passed through the solution, contain...A mixture upon adding conc. H2SO4 gives deep red fumes. It may contain the anions pair :...