Analytical ChemistryHard
Question
Which of the following compound is formed in borax bead test ?
Options
A.Orthoborate
B.Metaborate
C.Double oxide
D.Tetraborate
Solution
Na2B4O7.10H2O 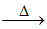 Na2B4O7 + 10H2O ; Na2B4O7
Na2B4O7 + 10H2O ; Na2B4O7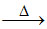 2NaBO2 + B2O3
2NaBO2 + B2O3
CuO + B2O3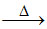 Cu(BO2)2 (copper (II) metaborate) - red and opaque bead in oxidising flame.
Cu(BO2)2 (copper (II) metaborate) - red and opaque bead in oxidising flame.
CuO + B2O3
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
Which of the following combines with Fe(II) ions to form a brown complex?...Select the incorrect statement(s)....Upon treatment with ammonical H2S , the metal ion that precipitates as a sulfide is -...A white crystalline substance dissolves in water. On passing H2S in this solution, a black precipitate is obtained. The ...In the precipitation of the iron group in qualitative analysis, ammonium chloride is added before adding ammonium hydrox...