s-Block ElementsHard
Question
Lime stone 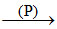 Quick lime
Quick lime 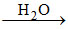
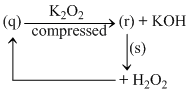
Which of the following option describes the correct reactants, products and reaction conditions.
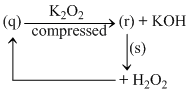
Which of the following option describes the correct reactants, products and reaction conditions.
Options
A.(p) Strongly heating (q) Slaked lime (r) Ca(O2)2 (s) Cold water
B.(p) Strongly heating (q) Slaked lime (r) CaO2 (s) Cold water
C.(p) Strongly heating in presence of catalyst (q) Slaked lime (r) CaO (s) Cold water
D.(p) Strongly heating under high pressure (q) Slaked lime (r) CaO2 (s) Cold water
Solution
CaCO3 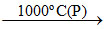 CaO
CaO 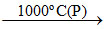
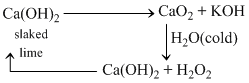
With hot water O2 is liberated.
CaO2 + H2O (hot) → Ca(OH)2 + O2

With hot water O2 is liberated.
CaO2 + H2O (hot) → Ca(OH)2 + O2
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
The following flow diagram represents the manufacturing of sodium carbonate ?2NH3 + H2O + CO2 → (NH4)2CO3 NH4HCO3 ...The ionization enthalpies of the alkali metals decrease down the group from Li to Cs because :...Which of the following has lowest thermal stability ?...Consider the following statements ;S1 : Beryllium and Magnesium are inert to oxygen and water. S2 : Concentrated solutio...Match the compounds /metal in (X) with their uses in (Y) : X YA. liquid sodium metal I breathing apparatus in submarineB...