s-Block ElementsHard
Question
The following flow diagram represents the manufacturing of sodium carbonate ?
2NH3 + H2O + CO2 → (NH4)2CO3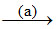 NH4HCO3
NH4HCO3 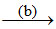
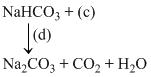
Which of the following option describes the underlined reagents, products and reaction conditions ?
2NH3 + H2O + CO2 → (NH4)2CO3
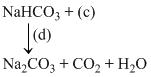
Which of the following option describes the underlined reagents, products and reaction conditions ?
Options
A.(a) Carbon dioxide, (b) NaCl (c) NH4Cl, (d) Heat
B.(a) Carbon dioxide, (b) NaCl (c) NH4Cl, (d) catalyst
C.(a) Higher tempt., (b) NaCl (c) NH4Cl, (d) Heat
D.(a) Higher pressure, (b) NaCl (c) NH4Cl, (d) Catalyst
Solution
NH3 + H2O + CO2 → (NH4)2CO3 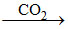 NH4HCO3
NH4HCO3
NH4HCO3 + NaCl → NaHCO3 + NH4Cl.
2 NaHCO3 Na2CO3 + CO2 + H2O.
Na2CO3 + CO2 + H2O.
NH4HCO3 + NaCl → NaHCO3 + NH4Cl.
2 NaHCO3
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Which one of the following electrolyte is used in Down′s process of extracting sodium metal ?...What happens when a mixture of sodium sulphate, lime stone and coke is heated in a furnace ?...When hydrated MgCl2. 6H2O is strongly heated :...Low solubility of CsI in water is due to :...Alkaline earth metal salts are :...