p-Block elementsHard
Question
KClO3 + H2SO4 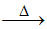 KHSO4 + HClO4 + (X) + H2O
KHSO4 + HClO4 + (X) + H2O
In above unbalanced chemical equation, the product (X) may be :
In above unbalanced chemical equation, the product (X) may be :
Options
A.O2
B.Cl2
C.ClO2
D.HCl
Solution
3KClO3 + 3H2SO4 → 2KHSO4 + HClO4 + 2ClO2 + H2O
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
When 8.3 g copper sulphate reacts with excess of potassium iodide then the amount of iodine liberated is :...Which of the following compound is obtained on heating potassium ferrocyanide with concentrated H2SO4?...The mixture of concentrated HCl and HNO3 made in 3 : 1radio contains :...Sulphur on oxidation with hot sulphuric acid gives :...Which is / are true statement(s) ?...