p-Block elementsHard
Question
Which is / are true statement(s) ?
Options
A.Basic nature of X- is in order F- > Cl- > Br- > I-
B.HI is strongest acid of HF, HCl, HBr and HI
C.The ionic character of M − X bond decreases in the order M − F > M − Cl > M − Br > M − I
D.Among F, Cl, Br and I, F has the highest enthalpy of hydration.
Solution
(A) Order is I- > Br- > Cl- > F- ; ease of donation of lone pair of electrons increases with increase in size of ions.
(B) HI is strongest acid on account of lowest bond dissociation energy.
(C) According to Fajans rule smaller anion has smaller polarisability and thus less covalent character but more ionic character.
(D) ᐃHHydration ∞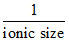
(B) HI is strongest acid on account of lowest bond dissociation energy.
(C) According to Fajans rule smaller anion has smaller polarisability and thus less covalent character but more ionic character.
(D) ᐃHHydration ∞
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
H3PO2 $\overset{\quad\Delta\quad}{\rightarrow}$ (X) + PH3 ; is...Identify the incorrect statement among the following...Crystalline boron in small amounts may be obtained by :...When borax is dissolved in water :...Which of the following does not have S − S linkage but have O − O linkage ?...