Ionic EquilibriumHard
Question
Number of H+ ions present in 10 mL of solution of pH = 3 are:
Options
A.1013
B.6.02 × 1018
C.6.02 × 1013
D.6.02 × 1010
Solution
Meq. of H+ = 10-3 × 10 = 10-2
∴ Number of H+ ions =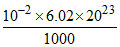 = 6.02 × 1018
= 6.02 × 1018
∴ Number of H+ ions =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Aqueous solutions of HNO3, KOH, CH3COOH, and CH3COONa of identical concentrations are provided. The pair (s) of solution...The pH of a 0.1 molar solution of the acid HQ is 3. The value of the ionization constant, Ka of this acid is :-...A solution contains 4.25 g ammonia per 250.0 ml of solution. Electrical conductivity measurement at 25°C shows that 0.40...Let the colour of the indicator HIn (colourless) will be visible only when its ionised form (pink) is 25% or more in a s...Which of the following salts undergoes anionic hydrolysis?...