Ionic EquilibriumHard
Question
Select the correct statements :
1. pH of NaHCO3 solution can be given by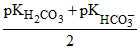
2. AI3+ ion is amphoteric
3. KSP values of metal nitrates are very-very high
4. Na+ (aq). is conjugate acid of NaOH (aq).
1. pH of NaHCO3 solution can be given by
2. AI3+ ion is amphoteric
3. KSP values of metal nitrates are very-very high
4. Na+ (aq). is conjugate acid of NaOH (aq).
Options
A.1, 2, 3, 4
B.1, 2, 4
C.2, 3, 4
D.1, 3, 4
More Ionic Equilibrium Questions
A sample of hard water contains 0.005 mole of CaCl2 per litre. What is the minimum concentration of Al2(SO4)3 which must...At infinite dilution the percentage dissociation of both weak acid and weak base is:...What is the pH at the equivalence point in a titration of 0.02 M NH3 with 0.02 M HBr. For the NH3, Kb = 1.8 × 10-5 ...The vapour pressure of a given liquid will decrease if....An example of a reversible reaction is :...