Ionic EquilibriumHard
Question
Equal volumes of following solutions are mixed, in which case the pH of resulting solution will be average value of pH of two solutions.
Options
A.Aqeous HCl of pH = 2, aqueous NaOH of pH = 12
B.Aqeous HCl of pH = 2, aqueous HCl of pH = 4
C.Aqeous NaOH of pH = 12, aqueous NaOH of pH = 10
D.Aqeous CH3COOH of pH = 5, aqueous NH3 of pH = 9. [Ka (CH3COOH) = Kb (NH3)]
Solution
(A) [H+] = 10-2 and [OH-] = 10-2
H+ + OH- → H2O
This leads complete neutralisation
So pH = 7 =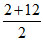
(D) [H+] = 10-5 and [OH-] = 10-5
[H+] + [OH-] H2O
This leads complete neutralisation
so pH = 7 =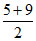
H+ + OH- → H2O
This leads complete neutralisation
So pH = 7 =
(D) [H+] = 10-5 and [OH-] = 10-5
[H+] + [OH-] H2O
This leads complete neutralisation
so pH = 7 =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
0.1mol HCl is dissolved in distilled water of volume V then at limV→∞ (pH)solution is equal to...A certain weak acid has a dissociation constant of 1.0 × 10-4. The equilibrium constant for its reaction with a str...How many grams of HCl should be dissolved in sufficient water to get 500 ml of an aqueous solution of pH, 2.0?...Which one is the correct graph (figure) for the corresponding acid base titration?...10-6 M HCI is diluted to 100 times. Its pH is:...