Ionic EquilibriumHard
Question
10-6 M HCI is diluted to 100 times. Its pH is:
Options
A.6.0
B.8.0
C.6.95
D.9.5
Solution
New concentration of; HCI = 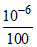 = 10-8 M
= 10-8 M
[H+] = 10-7 + 10-8 (approximately)
(Little less than 10-7 from water).
[H+] = 10-7 + 10-8 (approximately)
(Little less than 10-7 from water).
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
pH of 0.1M Na2HPO4 and 0.2M NaH2PO4 are respectively : pKa for H3PO4 are 2.2, 7.2 and 12.0....Common hydrolysis so products of XeF2 & XeF4 are :-...The solubility products of Al(OH)3 and Zn(OH)2 are 8.5 × 10-23 and 1.8 × 10-14 respectively. If NH4OH is added...To a 200 ml of 0.1 M weak acid HA solution 90 ml of 0.1 M solution of NaOH be added. Now, what volume of 0.1 M NaOH be a...20 cm3 of x M solution of HCl is exactly neutralised by 40 cm3 of 0.05 M NaOH solution, the pH of HCl solution is...