Ionic EquilibriumHard
Question
At 25oC, the solubility product values of AgCl and AgCNS are 1.8 × 10-10 and 1.6 × 10-11 respectively. When a solution is saturated with both solids, calculate the ratio [Cl-]/[CNS-] and also [Ag+] in the solution.
Options
A.1.125, 4 × 10-6 M
B.11.25, 1.4 × 10-5 M
C.1.25, 4 × 10-5 M
D.1.25, 4 × 10-6 M
Solution
AgCI → Ag+ → + CI- AgCNS → Ag+ + CNS-
(x + y) x (x + y) y
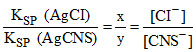 So
So 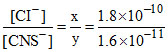
= 1.125 × 10 = 11.25
⇒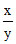 = 11.25 x = 11.25
= 11.25 x = 11.25
KSP (AgCI) = [Ag+] [CI-] = (x + y) x = 1.8 × 10-10
12.25 y × 11.25 y = 1.8 × 10-10
y2 =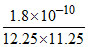
y2 =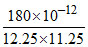
y2 = 1.3 × 10-12
y = 1.14 × 10-6
x = 11.25 × 1.14 × 10-6 = 12.83 × 10-6
[Ag+] = [x + y] = 12.83 × 10-6 + 1.14 × 10-6 = 13.97 × 10-6 = 1.4 × 10-5 M
(x + y) x (x + y) y
= 1.125 × 10 = 11.25
⇒
KSP (AgCI) = [Ag+] [CI-] = (x + y) x = 1.8 × 10-10
12.25 y × 11.25 y = 1.8 × 10-10
y2 =
y2 =
y2 = 1.3 × 10-12
y = 1.14 × 10-6
x = 11.25 × 1.14 × 10-6 = 12.83 × 10-6
[Ag+] = [x + y] = 12.83 × 10-6 + 1.14 × 10-6 = 13.97 × 10-6 = 1.4 × 10-5 M
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
When 0.1 mole solid NaOH is added in 1lt of 0.1M NH3(aq) then which statement is going to wrong?(Kb = 2 × 10-5, log...Degree of hydrolysis for a salt of strong acid and weak base is...Which is not true about acetaminophen ?...An acid with molelcular formula C7H6O3 forms three types of sodium salts. i.e., C7H5O3Na, C7H4O3Na2 and C7H3O3Na3. The b...The formation constant of Cu(NH3)42+ is 1.25 × 1012. What will be the equilibrium concentration of Cu2+ if 0.0125 moles ...