Ionic EquilibriumHard
Question
20 mL of a weak monobasic acid (HA) requires 20 mL 0.2 M NaOH for complete titration. If pH of solution upon addition of 10 mL of this alkali to 25 mL of the above solution of HA is 5.8. The pKa of the weak acid is
Options
A.6.1
B.5.8
C.5.98
D.5.58
Solution
meq. of acid = meq of base ⇒ 20 × M = 20 × 0.2 = 4
Molarity of HA = 0.2 M
HA + OH → A- + H2O
5 2 m. mole
3 - 2 m. mole
∴ pH = pKa + log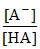 ⇒ 5.8 = pKa + log
⇒ 5.8 = pKa + log 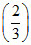 ⇒ pKa = 5.98
⇒ pKa = 5.98
Molarity of HA = 0.2 M
HA + OH → A- + H2O
5 2 m. mole
3 - 2 m. mole
∴ pH = pKa + log
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The reverse process of neutralisation is:...One mole of CH3COOH and one mole of C2H5OH reacts to produce mol of CH3COOC2H2. The equilibrium constant is:...Four species are listed below i. HCO3- ii. H3O+ iii. HSO4- vi. HSO3F Which one of the following is the correct sequence ...Solubility product of a salt AB is 1 × 10-8 in a solution in which th concentration of A+ ions is 10-3 M. The salt ...The electrolytic reduction of nitrobenzene in strongly acidic medium produces :-...