Ionic EquilibriumHard
Question
An acid-base indicator which is a weak acid has a pKIn value = 5.45. At what cocentration ratio of sodium acetate to acetic acid would the indicator show a colour half-way between those of its acid and conjugate base forms? [pKa of acetic acid = 4.75, log 2 = 0.3]
Options
A.4 : 1
B.6 : 1
C.5 : 1
D.3 : 1
Solution
pKa = 5.45
pH = pKHIn + log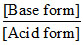 ⇒ pH = pKHIn = 5.45
⇒ pH = pKHIn = 5.45
For a Buffer solution
pH = pKa + log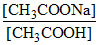 ⇒
⇒ 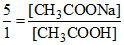
pH = pKHIn + log
For a Buffer solution
pH = pKa + log
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
What is the equilibrium constant of the following reaction? Fe(OH)3(s) + 3H3O+ $\rightleftharpoons$ Fe3+ + 6H2O?Ksp of ...When 0.1 mol arsenic acids (H3AsO4) is dissolved in 1L buffer solution of pH = 8, which of the following hold good? For ...Let the solubilities of AgCl in pure water, 0.01 M CaCl2, 0.01 M NaCl & 0.05 M AgNO3 be s1, s2, s3 & s4 respectively wha...The solubility of Li3Na3(AlF6)2 is 0.0744 g per 100 ml at 298 K. Calculate the solubility product of the salt (Atomic ma...For the titration of a dibasic weak acid H2A $\left( p^{K_{a(2)}} - p^{K_{a(1)}} \geq 2 \right)$ with a strong base, pH ...