Ionic EquilibriumHard
Question
Let the solubilities of AgCl in pure water, 0.01 M CaCl2, 0.01 M NaCl & 0.05 M AgNO3 be s1, s2, s3 & s4 respectively what is the correct order of these quantities. Neglect any complexation.
Options
A.s1 > s2 > s3 > s4
B.s1 > s2 = s3 > s4
C.s1 > s3 > s2 > s4
D.s4 > s2 > s3 > s1
Solution
Let Ksp of AgCI = x
(a) solubility of AgCl in pure water = s1 = √x (b) solubility of AgCl in 0.01 M CaCl2 = s2 =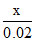
(c) solubility of AgCl in 0.01 M NaCl = s3 =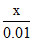 (d) solubility of AgCl in 0.05 M AgNO3 = s4 =
(d) solubility of AgCl in 0.05 M AgNO3 = s4 = 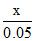
So s1 > s3 > s2 > s4
(a) solubility of AgCl in pure water = s1 = √x (b) solubility of AgCl in 0.01 M CaCl2 = s2 =
(c) solubility of AgCl in 0.01 M NaCl = s3 =
So s1 > s3 > s2 > s4
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
At 25o C, the dissociation constants of acid HA and base BOH in aqueous solution is same. The pH of 0.01 M solution of H...At what pH, is the solubility of Zn(OH)2 minimum? What is the minimum solubility?Zn(OH)2(s) $\rightleftharpoons$ Zn2+ (a...At 30oC the solubility of Ag2CO3 (KSP = 8 × 10-12) would be greatest in one litre of:...At infinite dilution the percentage dissociation of both weak acid and weak base is:...What is the conjugate base of OH- ?...