Ionic EquilibriumHard
Question
What fraction of an indicator HIn is in basic form at a pH of 6 if the pKa of the indicator is 5 ?
Options
A.1/2
B.1/11
C.10/11
D.1/10
Solution
pH = pKa + log 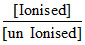 ⇒ 6 = 5 + log
⇒ 6 = 5 + log 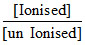 ⇒ 1 = log
⇒ 1 = log 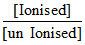 = 10 ⇒
= 10 ⇒ 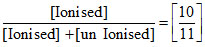
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A nucleotide consists of :-...How many moles of NaOH must be removed from one litre of aqueous solution to change its pH from 12 to 11 ?...Fear or excitement, generally cause on to breathe rapidly and it results in the decrease of CO2 concentration in blood. ...Which of the following solutions would have same pH?...Hydrogen ion concentration in mol / L in a solution of pH = 5.4 will be...