Ionic EquilibriumHard
Question
Which of the following solutions would have same pH?
Options
A.100 ml of 0.2 M HCl + 100 ml of 0.4 M NH3
B.50 ml of 0.1 M HCl + 50 ml of 0.2 M NH3
C.100 ml of 0.3 M HCl + 100 ml of 0.6 M NH3
D.All will have same pH.
Solution
(a) NH4OH + HCI → NH4CI + H2O
0.2 M 0.1 0 0
0.1 0 0.1 0.1
pOH = pKb
(b) NH4OH + HCI → NH4CI + H2O
0.1 0.05 0 0
0.05 0 0.05
pOH = pKb + log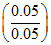 = pKb
= pKb
(c) NH4OH + HCI → NH4CI + H2O
0.3 0.15
0.15 0 0.15
pOH = pKb So all solution have same pH
0.2 M 0.1 0 0
0.1 0 0.1 0.1
pOH = pKb
(b) NH4OH + HCI → NH4CI + H2O
0.1 0.05 0 0
0.05 0 0.05
pOH = pKb + log
(c) NH4OH + HCI → NH4CI + H2O
0.3 0.15
0.15 0 0.15
pOH = pKb So all solution have same pH
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
When 20 mL of M/20 NaOH is added to 10 mL of M/10 HCl, the resulting solution will...Which one is the correct graph (figure) for the corresponding acid base titration?...The base imidazole has a Kb of 1.0 × 10–7 at 25°C. In what volumes should 0.02 M-HCl and 0.02 M imidazole be mixed to ma...Graphite is :-...pH of a mixture containing 0.10 M X- and 0.20 M HX is: [pKb (X-) = 4]...