Ionic EquilibriumHard
Question
When 100 ml of 0.4 M CH3COOH are mixed with 100 ml of 0.2 M NaOH, the [H3O+] in the solution is
approximately : [Ka(CH3COOH) = 1.8 × 10-5]
approximately : [Ka(CH3COOH) = 1.8 × 10-5]
Options
A.1.8 × 10-6 M
B.1.8 ×10-5 M
C.9 × 10-6 M
D.9 × 10-5 M.
Solution
CH3COOH + NaOH → CH3COONa + H2O
time t = 0 40 mmole 20 mmole
time t = t 20 mmole - 20 mmole
pH = pKa+ log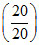 ⇒ pH = pKa ⇒ [H+] = Ka = 1.8 × 10-5 M
⇒ pH = pKa ⇒ [H+] = Ka = 1.8 × 10-5 M
time t = 0 40 mmole 20 mmole
time t = t 20 mmole - 20 mmole
pH = pKa+ log
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The equilibrium constant of T2O (Tritium is an isotope of H) differ from those of H2O at 298 K. Let at 298 K, pure T2O h...To a 200 ml of 0.1 M weak acid HA solution 90 ml of 0.1 M solution of NaOH be added. Now, what volume of 0.1 M NaOH be a...A aqueous solution contains an unknown concentration of Ba2+. When 50 mL of a 1 M solution of Na2SO4 is added, BaSO4 jus...Acetic acid and propionic acid have Ka values 1.75 × 10-5 and 1.3 × 10-5 respectively at a certain temperature...Select the correct order :-...