Ionic EquilibriumHard
Question
Acetic acid and propionic acid have Ka values 1.75 × 10-5 and 1.3 × 10-5 respectively at a certain temperature. An equimolar solution of a mixture, of the two acids is partially neutralised by NaOH. How is the ratio of the contents of acetate and propionate ions related to the Ka values and the molarity:
Options
A.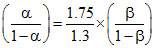 , where a and b are ionised fractions of their acids
, where a and b are ionised fractions of their acids
B.The ratio is unrelated to the Ka values.
C.The ratio is unrelated to the molarity of acid.
D.The ratio is unrelated to the pH of the solution.
Solution
CH3COOH ⇋ CH3COO- + H+ C2H5COOH ⇋ C2H5COO- + H+
1- α β α + β⎕⎕-x 1-β β α + β - x
∴ KA.A =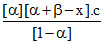 KP.A =
KP.A = 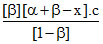
(where ′x′ is equivalents of NaOH droped).
∴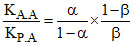 or
or 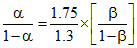
Hence, A,C,D.
1- α β α + β⎕⎕-x 1-β β α + β - x
∴ KA.A =
(where ′x′ is equivalents of NaOH droped).
∴
Hence, A,C,D.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A 0.1 M sodium acetate solution was prepared. The Kh = 5.6 × 10-10...Which of the following solution will have pH close to 1.0 ?...What is the aqueous ammonia concentration of a solution prepared by dissolving 0.15 mole of NH4+CH3COO− in 1 L of water?...A buffer solution is 0.25 M – CH3COOH + 0.15 M – CH3COONa, saturated in H2S (0.1 M) and has [Mn2+] = 0.04 M, Ka(CH3COOH)...Which may be added to one litre of water to act a buffer:...