Ionic EquilibriumHard
Question
A certain weak acid has a dissociation contant 1.0 × 10-4. The equilibrium constant for its reaction with a strong base is:
Options
A.1.0 × 10-4
B.1.0 × 10-10
C.1 × 10-10
D.1.0 × 10-14
Solution
HA ⇋ H+ + A- Ka = 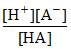 .....(i)
.....(i)
Also HA + B+ + OH- → B+ + A- + H2O Keq. =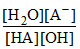 ......(ii)
......(ii)
By (i) and (ii),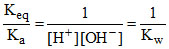 ∴ Keq. =
∴ Keq. = 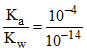 = 1010
= 1010
Also HA + B+ + OH- → B+ + A- + H2O Keq. =
By (i) and (ii),
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The value Kp for the reaction :2H2S(s) = 2H2(g) + S2(g) is 1.2 × 10-2 at 1065oC. The value for KC is :...Degree of hydrolysis for a salt of strong acid and weak base is...A solution having hydrogen ion concentration is 0.0005 g eqvt./litre, its pOH is :...To a 200 ml of 0.1 M weak acid HA solution 90 ml of 0.1 M solution of NaOH be added. Now, what volume of 0.1 M NaOH be a...In water, the acid HCIO4. HCI, H2SO4 and HNO3 exhibit the same strength as they are completely ionised in water (a base)...