General Organic ChemistryHard
Question
Which one is most reactive towards SN1 reaction ?
Options
A.C6H5CH(C6H5)Br
B.C6H5CH(CH3)Br
C.C6H5C(CH3)(C6H5)Br
D.C6H5CH2Br
Solution
SN1 reaction involves the formation of carbocation intermediate. More the stability of carbocation, more is the reactivity of alkyl/aryl halides towards SN1 reaction.
The intermediate carbocation formed by given halides are as :
(a) C6H5CH(C6H5) Br → (C6H5)2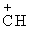 + Br-
+ Br-
(b) C6H5CH(CH3)Br → C6H5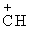 (CH3) + Br-
(CH3) + Br-
(c) C6H5C(CH3)(C6H5)Br → C6H5)2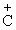 (CH3) + Br-
(CH3) + Br-
(d)C6H5CH2Br → (C6H5)2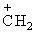 + Br-
+ Br-
The order of stability of these carbocations is as
(C6H5)2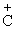 (CH3) > (C6H5)2
(CH3) > (C6H5)2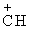 > C6H5
> C6H5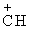 (CH3) > (C6H5)2
(CH3) > (C6H5)2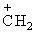
Thus, C6H5C(CH3)(C6H5)Br is most reactive towards SN1 reaction.
The intermediate carbocation formed by given halides are as :
(a) C6H5CH(C6H5) Br → (C6H5)2
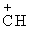 + Br-
+ Br- (b) C6H5CH(CH3)Br → C6H5
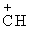 (CH3) + Br-
(CH3) + Br-(c) C6H5C(CH3)(C6H5)Br → C6H5)2
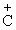 (CH3) + Br-
(CH3) + Br-(d)C6H5CH2Br → (C6H5)2
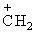 + Br-
+ Br- The order of stability of these carbocations is as
(C6H5)2
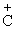 (CH3) > (C6H5)2
(CH3) > (C6H5)2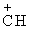 > C6H5
> C6H5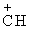 (CH3) > (C6H5)2
(CH3) > (C6H5)2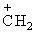
Thus, C6H5C(CH3)(C6H5)Br is most reactive towards SN1 reaction.
Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
The hybridisation of carbon atoms in C - C single bond of H - C ≡ C - CH = CH2 is :...Consider the following bromides : The correct order of SN1 reactivity is...Which of the the following is optically inactive ?...The IUPAC of following compound is...Which of the following has the highest nucleophilicity ?...