General Organic ChemistryHard
Question
Which of the following has the highest nucleophilicity ?
Options
A.F-
B.OH-
C.CH3-
D.NH2-
Solution
Nucleophilicity represents the capacity of a Nu>- to attack on electron deficient centre, when comparing the nucleophilicity of species in which attacking atom belongs to the same row of periodie table nucleopilicity is roughly in the order og their basicity and order of basic strength (nucleopilicity)
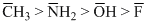
because corresponding acids follow the order of their acidic strength as :
CH4 < NH3 O2H < HF
Note that the acidic character of hydrides in a period increases on moving from left to fight.
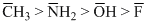
because corresponding acids follow the order of their acidic strength as :
CH4 < NH3 O2H < HF
Note that the acidic character of hydrides in a period increases on moving from left to fight.
Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
In which of the following does the C−H bond (shown by a thick line) have the least bond dissociation energy -...The meta-directing power of the groups −NH2, −OCH3, −C6H5 and − NO2 follows the order...An SN2 reaction of an asymmetric carbon of a compound always gives :...Which of the following is the strongest base -...Arrange acidity of given compounds in decreasing order :(I) CH3-NH-CH2-CH2-OH(II) CH3-NH-CH2-CH2-CH2-OH (III) (CH3)3N+ -...