Ionic EquilibriumHard
Question
A 0.1 M sodium acetate solution was prepared. The Kh = 5.6 × 10-10
Options
A.The degree of hydrolysis is 7.48 × 10-5
B.The [OH-] concentration is 7.48 × 10-3 M
C.The [OH-] concentration is 7.48 × 10-6 M
D.The pH is approximately 8.88.
Solution
CH3COO- + H2O CH3COOH + OH-.
0.1 (1 - h) 0.1 h 0.1 h.
Kh =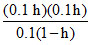 = 0.1 h2 ⇒ 5.6 × 10-10 = 0.1 h2
= 0.1 h2 ⇒ 5.6 × 10-10 = 0.1 h2
⇒ h = 7.48 × 10-5 [∴ h <<<< 1]
[OH-] = ch = 7.48 × 10-5 × 10-1 = 7.48 × 10-6.
[H+] =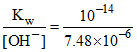 = 1.33 × 10-9.
= 1.33 × 10-9.
⇒ pH = 8.8 approx.
0.1 (1 - h) 0.1 h 0.1 h.
Kh =
⇒ h = 7.48 × 10-5 [∴ h <<<< 1]
[OH-] = ch = 7.48 × 10-5 × 10-1 = 7.48 × 10-6.
[H+] =
⇒ pH = 8.8 approx.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
M(OH)x has KSP 4 × 10-12 and solubility 10-4 M. The value of x is:...When 100 ml of 0.4 M CH3COOH are mixed with 100 ml of 0.2 M NaOH, the [H3O+] in the solution is approximately : [Ka(CH3C...The indicator constant for an acidic indicator, HIn is 5 × 10−6 M. This indicator appears only in the colour of acidic f...Calculate the pH of 0.02 M – HA solution. The value of Ka for HA = 2 × 10−12 (log 2 = 0.3, log 3 = 0.48)....Choose the correct statement:...