Ionic EquilibriumHard
Question
For two different acids with same concentration
Options
A.the relative strength is expressed as 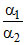
B.relative strength is expressed as 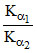
C.relative strength is expressed as 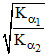
D.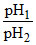
Solution
Let α1 and α2 be the degree of dissociation of two acids and α1, α2 are very small when compared to unity.
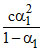 = Ka1
= Ka1 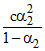 = Ka2
= Ka2
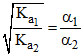 = relative strength.
= relative strength.
Hence choices (A) and (C) are correct while (B) and (D) are incorrect.
Hence choices (A) and (C) are correct while (B) and (D) are incorrect.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Which of the following compounds is not an antacid?...The acidic hydrolysis of ether (X) shown below is fastest when   Â...The amino acid glycine (NH2CH2COOH) is basic because of its –NH2 group and acidic because of its –COOH group. By a proce...pH of 0.01 M HS- will be:...pOH of H2O is 7.0 at 298 K. If water is heated at 350 K, which of the following statement should be true?...