Ionic EquilibriumHard
Question
Which of the following solution will have pH close to 1.0 ?
Options
A.00 ml of M/10 HCl + 100 ml of M/10 NaOH
B.55 ml of M/10 HCl + 45 ml of M/10 NaOH
C.10 ml of M/10 HCl + 90 ml of M/10 NaOH
D.75 ml of M/5 HCl + 25 ml of M/5 NaOH.
Solution
(a) HCI NaOH
No. of mili eq. =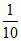 × 100 = 10
× 100 = 10 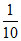 × 100 = 10
× 100 = 10
So solution is Neutral
(b)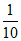 × 55 = 5.5
× 55 = 5.5 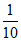 × 45 = 4.5
× 45 = 4.5
[H+] =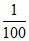 = 10-2 M, pH = 2
= 10-2 M, pH = 2
(c)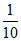 × 10 = 1
× 10 = 1 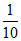 × 90 = 9 Basic
× 90 = 9 Basic
(d)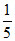 × 75 = 15
× 75 = 15 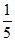 × 25 = 5
× 25 = 5
[H+] = 0.1 M, pH = 1
No. of mili eq. =
So solution is Neutral
(b)
[H+] =
(c)
(d)
[H+] = 0.1 M, pH = 1
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Equal volumes of equimolar solutions of a weak acid and a weak base both having dissociation constant equal to 5 × ...For pure water...A volume of 50 ml of a solution which is 0.05 M in the acid HA (pKa = 3.80) and 0.08 M in HB (pKa = 8.20) is titrated wi...pH at which a basic indicator with Kb = 1.0 × 10-10 changes colour when the concentration of indicator is 10-2 M :-...XeF4 + O2F2 → [X] + O2Select the correct statement(s) for [X]...