Ionic EquilibriumHard
Question
The concentration of hydroxyl ion in a solution left after mixing 100 mL of 0.1 M MgCI2 and 100 mL of 0.2 M NaOH (KSP of Mg(OH)2 = 1.2 × 10-11):
Options
A.2.88 × 10-3
B.2.88 × 10-2
C.2.88 × 10-4
D.2.88 × 10-5
Solution
MgCl2 + 2NaOH → Mg(OH)2 + 2NaCl
mm before 10 20 0 0
reaction 0 0 10 20
Thus, 10 m mole of Mg(OH)2 are formed. The product of [Mg2+][OH-]2 is therefore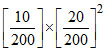 = 5 √ 10-4 which is more than KSP of Mg(OH)2. Now solubility(s) of Mg(OH)2 can be derived by
= 5 √ 10-4 which is more than KSP of Mg(OH)2. Now solubility(s) of Mg(OH)2 can be derived by
KSP = 4s3
∴ s =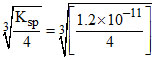 = 1.4 × 10-4 M
= 1.4 × 10-4 M
∴ [OH-] = 2s = 2.88 × 10-4 M
mm before 10 20 0 0
reaction 0 0 10 20
Thus, 10 m mole of Mg(OH)2 are formed. The product of [Mg2+][OH-]2 is therefore
KSP = 4s3
∴ s =
∴ [OH-] = 2s = 2.88 × 10-4 M
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
In which of the following reactions, the concentration of the product is hither than theconcentration of the product is ...At 25oC, the solubility product of Mg(OH)2 is 1.0 × 10-11. At which pH, will Mg2+ ions start precipitating in the f...If valence factor (n-factor) of compound NaHC2O4.2H2 C2O4 .3K2C2O4. 4Al2(C2O4)3.3FeC2O4 in acid base titration is x and ...Solubility of BaF2 in a solution of Ba(NO3)2 will be represented by the concentration term:...Arsenic (III) sulphide form a sol with negative charge. Which of the following ionic substances will have lowest floceul...