Ionic EquilibriumHard
Question
The composition of an acidic buffer mixture made up of HA and NaA of total molarity 0.29 having pH = 4.4 and Ka = 1.8 × 10-5 in terms of concentration of salt and acid respectively is :
Options
A.0.09 M and 0.20 M
B.0.20 M and 0.09
C.0.1 M and 0.19 M
D.0.19 M and 010 M
Solution
pH = - log Ka + log 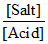
Let a mol litre-1 be concentration of salt, then concentration of acid = (0.29 - a).
4.4 = - log 1.8 × 10-5 + log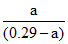
∴ a = 0.09.
[Salt] = 0.09 M
[Acid] = 0.29 - 0.09 = 0.20 M.
Let a mol litre-1 be concentration of salt, then concentration of acid = (0.29 - a).
4.4 = - log 1.8 × 10-5 + log
∴ a = 0.09.
[Salt] = 0.09 M
[Acid] = 0.29 - 0.09 = 0.20 M.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
When 20 ml of 0.2 M-DCl solution is mixed with 80 ml of 0.1 M-NaOD solution, pH of the resulting solution becomes 13.6. ...The pH of blood does not appreciably caange by asmnall addition of acid or a basebeacause blood....A buffer solution is prepared by mixing ′a′ moles of CH3COONa and ′b′ moles of CH3COOH such that...0.1mol HCl is dissolved in distilled water of volume V then at limV→∞ (pH)solution is equal to...Which of the following solution will have pH close to 1.0 ?...