Ionic EquilibriumHard
Question
10 mL of 10-6 M HCI solution is mixed with 90 mL H2O. pH will change approximately:
Options
A.By one unit
B.By 0.3 unit
C.By 0.7 unit
D.By 0.1 unit
Solution
pH of 10-6 M HCI = 6; on dilution [HCI] =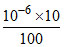 = 10-7
= 10-7
Thus new pH of HCI is not 7 because it is acid. For new solution pH:
[H+] = 10-7 + 10-7 (from H2O) = 2 × 10-7 ∴ pH = 6.7
(approximately)
Thus new pH of HCI is not 7 because it is acid. For new solution pH:
[H+] = 10-7 + 10-7 (from H2O) = 2 × 10-7 ∴ pH = 6.7
(approximately)
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
What will be the resultant pH when 200 ml of an aqueous solution of HCl (pH = 2.0) is mixed with 300 ml of an aqueous so...Which of following statements is false :-...KSP if Mg(OH)2 is 1 × 10-12. 0.01M MgCI2 will show precipitation in a solution of pH greater than:...Which of the following mixtures will act as buffer ?...If the solubility of lithium sodium hexafluorido aluminate, Li3Na3 (AlF6)2 is ′s′ mol lt-1, its solubility p...