Ionic EquilibriumHard
Question
Ka for the acid HA is 1 × 10-6. The value of K for the reaction A- + H3O+ ⇋ HA + H2O is
Options
A.1 × 10-6
B.1 × 1012
C.1 × 10-12
D.1 × 106
Solution
Ka = 10-6 for HA + H2O ⇋ H3O+ + A- Thus K for reverse reaction is 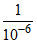 = 106
= 106
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The following graph represents the titration of pH vs volume Find out the best possible statement from the graph shown....A student was given 0.01 mole of a weak organic acid and told to determine the Ka of the acid. He prepared 100 ml of an ...50 mL of 0.1 M NaOH is added to 60 mL of 0.15 M H3PO4 solution (K1, K2 and K3 for H3PO4 are 10-3, 10-8 and 10-13 respect...After solid SrCO3 was equilibrated with a buffer at pH 8.6, the solution was found to have [Sr2+] = 2.0 × 10−4 M, what i...Four species are listed below i. HCO3- ii. H3O+ iii. HSO4- vi. HSO3F Which one of the following is the correct sequence ...