Mole ConceptHard
Question
25.3 g of sodium carbonate, Na2CO3 is dissolved in enough water to make 250 mL of solution. If sodium carbonate dissociates completely, molar concentration of sodium ion, Na+ and carbonate ion,CO32- are respectively
(Molar mass of Na2CO3 = 106 g mol-1)
(Molar mass of Na2CO3 = 106 g mol-1)
Options
A.0.955 M and 1.910 M
B.1.910 M and 0.955 M
C.1.90 M and 1.910 M
D.0.477 M and 0.477 M
Solution
Molarity =  × 1000
× 1000
=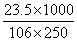 = .9547
= .9547 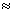 0.955 M
0.955 M
Na2CO3 in aqueous solution remains dissociated as
Na2CO3 ⇋ 2Na+ + CO32-
x 2x x
Since. the molarity of Na2CO3 is 0.955 M, the molarity of CO32- is also 0.95 M and that of Na+ is 2 × 0.955 = 1.910 M
 × 1000
× 1000=
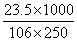 = .9547
= .9547 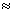 0.955 M
0.955 MNa2CO3 in aqueous solution remains dissociated as
Na2CO3 ⇋ 2Na+ + CO32-
x 2x x
Since. the molarity of Na2CO3 is 0.955 M, the molarity of CO32- is also 0.95 M and that of Na+ is 2 × 0.955 = 1.910 M
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Mole fraction of the solute in a 1.00 molal aqueous solution is...Two solutions of a substance (non electrolyte) are mixed in the following manner. 480 ml of 1.5 M first solution + 520 m...100 mL of 0.06 M Ca(NO3)2 is added to 50 mL of 0.06 M Na2C2O4. After the reaction is complete....2SO2 + O2 ⇋ 2SO3 initially 4 moles each of SO2 & O2 are present, at equilibrium 25% of O2 is used. Total mols at e...Given the polymers (i) Nylon 6,6 (ii) Buna-S, (iii) Polythene, arrange these in increasing order of their inter-molecula...