Mole ConceptHard
Question
100 mL of 0.06 M Ca(NO3)2 is added to 50 mL of 0.06 M Na2C2O4. After the reaction is complete.
Options
A.0.003 moles of calcium oxalate will get precipitated
B.0.003 M of Ca2+ will remain in excess
C.Na2C2O4 is limiting reagent.
D.Ca(NO3)2 is excess reagent.
Solution
(A), (C) and (D) Explanation :
Ca2+ + C2O42- → CaC2O4
100 ml of 0.06 M Ca(NO3)2 contains 100 × 0.06 = 6 millimole
50 ml of 0.06 M Na2C2O4 contains 50× 0.06 = 3 millimole
3 millimoles of C2O42- will react with 3 millimoles of Ca2+ for 3 millimoles of CaC2O4
=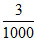 = 0.003 moles
= 0.003 moles
(A) is correct.
Excess of Ca2+ left = 6 - 3 = 3 millimoles =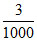 = 0.003 moles.
= 0.003 moles.
(C) is correct because sodium oxalate is present in smaller amount than required.
(D) is correct because Ca(NO3)2 is present in large amount.
Ca2+ + C2O42- → CaC2O4
100 ml of 0.06 M Ca(NO3)2 contains 100 × 0.06 = 6 millimole
50 ml of 0.06 M Na2C2O4 contains 50× 0.06 = 3 millimole
3 millimoles of C2O42- will react with 3 millimoles of Ca2+ for 3 millimoles of CaC2O4
=
(A) is correct.
Excess of Ca2+ left = 6 - 3 = 3 millimoles =
(C) is correct because sodium oxalate is present in smaller amount than required.
(D) is correct because Ca(NO3)2 is present in large amount.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
V1 ml of unknown gas (A) + V2 ml of O2 → (V1 + V2) ml of CO2. Gas ‘A’ may be...At 373 K and 1 atm, if the density of liquid water is 1.0 g/ml and that of water vapour is 0.0006 g/ml, then the volume ...How many litres of detonating gas may be produced at 0o C and 1 atm from the decomposition of 0.1 mole of water by an el...Vapour density of a volatile substance is 1.2 (C2H6 = 1). Its molecular mass would be...Law of multiple proportions is not applicable for the oxide(s) of...