Ionic EquilibriumHard
Question
A certain buffer solution contains equal concentration of X- and HX. The Kb for X- is 10-10. The pH of the buffer is:
Options
A.4
B.7
C.10
D.14
Solution
pH = - log Ka + log 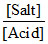 = - log 10-4 + log
= - log 10-4 + log 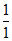 = 4
= 4
[Since Ka × Kb = 10-14 Given Kb = 10-10 ∴ Ka = 10-4]
[Since Ka × Kb = 10-14 Given Kb = 10-10 ∴ Ka = 10-4]
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
is converted in acetophenone by : -...The pH of a solution obtained by mixing 100 ml of 0.2 M CH3COOH with 100 ml of 0.2 M NaOH would be : (pKa for CH3COOH = ...10-6 M HCI is diluted to 100 times. Its pH is:...Which of the following when added to 1.0 L of 0.5 M HCl would result in maximum increase in pH :-...The species present in solution when CO2 is dissolved in water are...