Ionic EquilibriumHard
Question
The pH of a solution obtained by mixing 100 ml of 0.2 M CH3COOH with 100 ml of 0.2 M NaOH would be :
(pKa for CH3COOH = 4.74)
(pKa for CH3COOH = 4.74)
Options
A.4.74
B.8.87
C.9.10
D.8.57
Solution
CH3COOH + OH- CH3COO- + H2O
t = 0 20 20
t = eq - - 20
So, [CH3COO-] =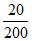 = 0.1 M
= 0.1 M
pH = 7 +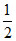 pKa +
pKa + 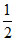 log C = 7 + 2.37 +
log C = 7 + 2.37 + 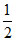 log 10-1 = 7 + 2.37 - 0.5 = 8.87
log 10-1 = 7 + 2.37 - 0.5 = 8.87
t = 0 20 20
t = eq - - 20
So, [CH3COO-] =
pH = 7 +
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
100 ml of pH = 6 (acidic) is diluted to 1000 ml by H2O. pH will increase by :-...Species acting as both Bronsted acid and base is...50 mL of 0.1 M NaOH is added to 75 mL of 0.1M NH4Cl to make a basic buffer. If pKa of NH4+ is 9.26 then pH will be :-...The dissociation constant of a weak monoprotic acid is numerically equal to the dissociation constant of its conjugate b...In the manufacture of NH3 in Haber′s continuous flow process involving the reaction H2(g) + 3H2(g) 2NH3(g) ᐃ...