Ionic EquilibriumHard
Question
When pure water is saturated with CaCO3 and CaC2O4, the concentration of calcium ion in the solution under equilibrium is 8 × 10-5 M. If the ratio of the solubility product of CaCO3 to that of CaC2O4 is 3, what is the solubility product of CaCO3 in pure water?
Options
A.4.80 × 10-8
B.9.60 × 10-9
C.9.60 × 10-8
D.4.80 × 10-9
Solution
CaCO3 ⇋ Ca+2 + CO3-2 CaC2O4 ⇋ Ca+2 + C2O4-2
x x y y
(x+y) x (x+y) y
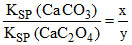 = 3 ⇒ x = 3y
= 3 ⇒ x = 3y
(x + y) = 8 × 10-5
(3y + y) = 8 × 10-5 ⇒ y = 2 × 10-5 x = 6 × 10-5
KSP (CaCO3) = [Ca+2] [CO3-2]
= 8 × 10-5 × 6 × 10-5
= 48 × 10-10
KSP (CaCO3) = 4.8 × 10-9
x x y y
(x+y) x (x+y) y
(x + y) = 8 × 10-5
(3y + y) = 8 × 10-5 ⇒ y = 2 × 10-5 x = 6 × 10-5
KSP (CaCO3) = [Ca+2] [CO3-2]
= 8 × 10-5 × 6 × 10-5
= 48 × 10-10
KSP (CaCO3) = 4.8 × 10-9
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
To a 200 ml of 0.1 M weak acid HA solution 90 ml of 0.1 M solution of NaOH be added. Now, what volume of 0.1 M NaOH be a...The solubility of Ag2CO3 in water at 25oC is 1 × 10-4 mole/litre. What is its solubility in 0.01 M Na2CO3 solution?...The self ionisation constant for pure formic acid, K = [HCOOH2+][HCOO-] has been estimated as 10-6 at room temperature. ...The precipitate of CaF2 (Ksp = 1.7 × 10-10) is obtained, when equal volumes of the following are mixed :...Let the solubility of an aqueous solution of Mg(OH)2 be x then its ksp is...